Order this document
by MPX2300DT1/D
SEMICONDUCTOR TECHNICAL DATA
PRESSURE SENSORS
Motorola has developed a low cost, high volume, miniature pressure sensor package
which is ideal as a sub–module component or a disposable unit. The unique concept of
the Chip Pak allows great flexibility in system design while allowing an economic solution
for the designer. This new chip carrier package uses Motorola’s unique sensor die with its
patented, piezoresistive implant technology, along with the added feature of on–chip,
thin–film temperature compensation and calibration.
Features:
•
•
Low Cost
Patented piezoresistive strain gauge implant, temperature compensation and
calibration all integrated on a single, monolithic sensor die.
CHIP PAK ELEMENT
CASE 423A–02
•
•
Pressure Range Available: 0–300 mmHg
Polysulfone (Mindell S–1000) Case Material (Medical, Class VI Approved)
Motorola is offering the Chip Pak option package. Application–specific parts will have
PIN NUMBER
an “SPX” prefix, followed by a four digit number, unique to the specific customer. Devices
will be shipped in a tape and reel packaging.
NOTE: The die and wire bonds are exposed on the front side of the Chip Pak (pressure
is applied to the backside of the device). Front side die and wire protection must be
provided in the customer’s housing. Use caution when handling the devices during all
processes.
1
2
V
3
4
S–
S
S+
Gnd
MAXIMUM RATINGS
Rating
Symbol
Value
125
Unit
PSI
kPa
Vdc
°C
Overpressure (Backside)
Burst Pressure
P
max
P
burst
1000
Supply Voltage
V
Smax
10
Storage Temperature
Operating Temperature
T
stg
–25 to +85
+15 to +40
T
A
°C
Motorola’s MPX2300DT1 Pressure Sensors. Motorola’s
MPX2300DT1 pressure sensor has been designed for
medical usage by combining the performance of Motorola’s
shear stress pressure sensor design and the use of
biomedically approved materials. Materials with a proven
history in medical situations have been chosen to provide a
sensor that can be used with confidence in applications,
such as invasive blood pressure monitoring. It can be
sterilized using ethylene oxide. The portions of the pressure
sensor that are required to be biomedically approved are the
rigid housing and the gel coating.
A silicone dielectric gel that has been used extensively in
implants covers the silicon piezoresistive sensing element.
The gel is a nontoxic, nonallergenic polymer system which
passes pyrogen testing, as well as meeting all USP XX Bio-
logical Testing Class VI requirements. The properties of the
gel allow it to transmit pressure uniformly to the diaphragm
surface, while isolating the internal electrical connections
from the corrosive effects of fluids, such as saline solution.
The gel provides electrical isolation sufficient to withstand
defibrillation testing, as specified in the proposed Association
for the Advancement of Medical Instrumentation (AAMI)
Standard for blood pressure transducers. A biomedically ap-
proved opaque filler in the gel prevents bright operating room
lights from affecting the performance of the sensor.
The rigid housing is molded from a white, medical grade
polysulfone that has passed extensive biological testing
including: tissue culture test, rabbit implant, hemolysis,
intracutaneous test in rabbits, and system toxicity, USP.
Senseon and X–ducer are trademarks of Motorola, Inc.
Motorola Sensor Device Data
1
Motorola, Inc. 1997






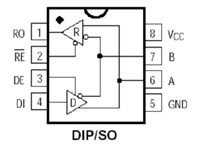 MAX487芯片引脚图及功能、应用领域详解
MAX487芯片引脚图及功能、应用领域详解

 IR2110驱动芯片引脚图及功能、电路图详解
IR2110驱动芯片引脚图及功能、电路图详解

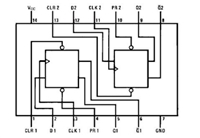 74LS74是什么芯片 74LS74引脚图及功能表
74LS74是什么芯片 74LS74引脚图及功能表

 CD4511芯片引脚图及功能、电路图解析
CD4511芯片引脚图及功能、电路图解析
